Proteinase K
Widely utilized in DNA, RNA and Phage Purification
Proteinase K is a serine protease that is used to digest proteins and remove result altering impurities from nucleic acid preparations. Proteinase K is a stable S8 family serine alkaline protease containing two disulfide bridges and one free Cys near His at the active site. The main site of cleavage is the peptide bond next to the carboxyl group of aliphatic and aromatic amino acids with blocked alpha amino groups.
In molecular biology research, adding Proteinase K to nucleic acid preparations inhibits nucleases that could metabolize DNA or RNA during purification and isolation experiments
Applications
- Many wide applications in molecular biology, cell biology, genomic DNA extraction, cell permeability, enzyme digestion and removal experiments as well as various more.
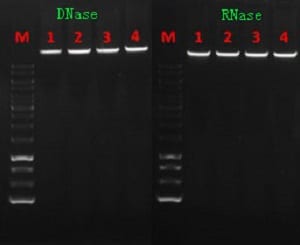
- DNA, RNA, and phage purification (Proteinase K is DNASE and RNase Free)
- Protein Digestion
- Proteinase K is widely utilized in DNA preparation for many processes such as: hybridization, fingerprinting, colony or plaque hybridization, and pulse-field electrophoresis.
Proteinase K: Activity, pH Range & Optimum Temperature
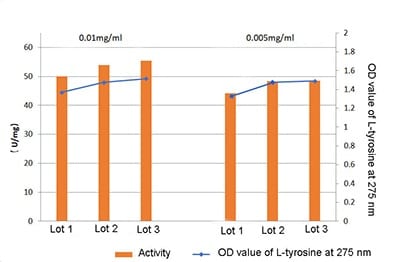
Proteinase K activity can be measured using haemoglobin as the substrate. At 37°C the amount of Proteinase K that can produce amino acids or polypeptides equivalent to 1 micromol of tyrosine Folin positive in one minute with haemoglobin as the substrate is defined as a unit of Proteinase K activity. Enzyme activity > 30U/mg.
The effective pH range of Proteinase K is approximately between pH4.0-12.5, and the optimal pH range is pH7.5-8.0.
The optimum reaction temperature of Proteinase K is 65°C, but quickly denatures after surpassing 65°C – The optimum temperature for Proteinase K reaction efficiency is between 50~55 °C.
Product Information Table
| Name | Packsize | Order | |
|---|---|---|---|
| We couldn't find any records. | |||